Medical Equipment Government Tenders: Quality Certification and Compliance Guide

Public Procurement in Medical Equipment Faces a New Era of Compliance Complexity
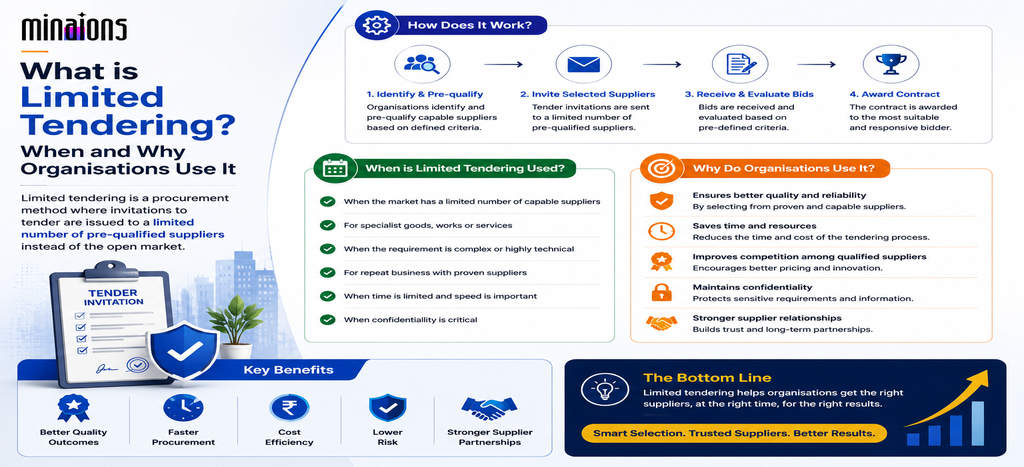
As healthcare systems worldwide confront supply chain fragility and rising patient safety expectations, public procurement teams now assess medical equipment beyond price and delivery timelines. Modern government tenders require verifiable evidence of quality certification, regulatory alignment, and ongoing compliance readiness. Failure to meet these standards risks disqualification, legal exposure, reputational damage, and threats to public health. Organisations treating compliance as a static checklist are losing contracts. Those integrating compliance into operational processes through intelligent systems secure tenders and build trusted partnerships with government agencies. The transition from transactional procurement to value-based acquisition is underway, with success favouring those who anticipate regulatory change before it becomes mandatory.
Essential Quality Certifications for Medical Equipment in Government Tenders
Winning medical equipment tenders demands more than technical specifications, it requires demonstrable adherence to globally recognised quality and safety standards. The cornerstone of compliance is ISO 13485, the international standard for quality management systems specific to medical devices. Regulatory bodies, including the U.S. Food and Drug Administration, are increasingly aligning their frameworks with ISO 13485 principles, making it a de facto baseline for eligibility in federal and state-level procurements. Equally critical is CE Marking, mandated under the EU Medical Device Regulation (EU MDR 2017/745), which certifies that devices meet essential health, safety, and environmental requirements for sale within the European Economic Area. For suppliers targeting U.S. federal contracts, FDA approval or clearance is non-negotiable, particularly for Class II and III devices. These certifications are not interchangeable; each serves a distinct jurisdiction and regulatory philosophy, requiring manufacturers to tailor their quality systems accordingly.
Additional standards such as Good Manufacturing Practices (GMP), IEC 60601 for electrical safety, and ISO 14971 for risk management further define the compliance landscape. Suppliers must maintain documented evidence of conformity for each, including design controls, production records, and post-market surveillance data. In public procurement, this documentation is not merely submitted, it is subject to rigorous audit by contracting officers and independent assessors. A single gap in traceability can invalidate an entire bid, regardless of technical merit.
The Strategic Role of Regulatory Frameworks in Tender Success
Compliance now extends beyond the factory floor into the procurement lifecycle, from pre-bid qualification to post-award contract management. The Trade Agreements Act (TAA) governs sourcing for U.S. federal procurements, requiring medical equipment to originate from designated countries. This impacts global manufacturers who must verify their supply chains against TAA-compliant nations, a task complicated by fragmented sourcing networks. Similarly, the Medical Device Single Audit Program (MDSAP) enables manufacturers to satisfy regulatory requirements across multiple jurisdictions, including Australia, Brazil, Canada, Japan, and the United States, through a single audit based on ISO 13485. This reduces duplication but demands a unified quality management structure.
Procurement agencies increasingly use outcome-based metrics, evaluating whether devices improve clinical outcomes, reduce hospital-acquired infections, or enhance supply continuity. This elevates the importance of post-market data, clinical evaluations, and real-world performance reporting, all of which must be embedded within the manufacturer’s quality system from the outset. Organisations treating compliance as a one-time submission, rather than an ongoing operational discipline, are excluded from future tenders.
AI-Powered Compliance as a Strategic Advantage in Public Procurement
The administrative burden of managing multiple certifications, regulatory updates, and tender-specific requirements creates a critical bottleneck for suppliers. Manual processes are slow, error-prone, and ill-equipped for the dynamic nature of global regulations. Here, the integration of Agentic AI solutions into the procurement workflow is transforming competition. These systems autonomously cross-reference tender documents against certified product data, flagging mismatches in certification scope, expiry dates, or regulatory jurisdiction before submission. They also monitor regulatory changes in real time, alerting teams to updates in EU MDR, FDA guidance, or TAA country lists, ensuring continuous preparedness.
Platforms leveraging AI for document automation extract key compliance evidence from technical files, clinical evaluations, and quality manuals, reducing submission preparation time from weeks to days. This capability is vital for smaller suppliers and specialist manufacturers lacking dedicated compliance teams. By automating verification, audit trail generation, and multilingual document alignment, these technologies enable organisations to compete with larger enterprises on equal footing. The result is faster, more accurate, defensible, and strategically aligned bids that align with modern public procurement priorities. Minaions supports this transformation through enterprise-grade AI systems designed for public sector compliance. Minaions enables suppliers to meet evolving regulatory demands with precision and scale.
Common Compliance Pitfalls and How to Avoid Them
Many suppliers fail in government tenders due to procedural missteps, not poor product quality. Common errors include submitting expired certification documents, misclassifying device risk levels under EU MDR, or assuming FDA clearance applies universally across all federal agencies. Another frequent oversight is neglecting data sovereignty requirements, particularly when using cloud-based systems for document storage or AI processing. Public sector agencies increasingly mandate that sensitive health data remain within national borders, requiring compliance solutions to be architecturally aligned with local regulations.
Additionally, organisations often treat certifications as isolated assets rather than integrated components of a broader compliance ecosystem. ISO 13485 must be linked to GMP, risk management, and post-market surveillance processes to form a cohesive quality framework. Without this integration, audits reveal fragmented systems that appear reactive rather than proactive. The most successful suppliers now design their compliance architecture around the principle of continuous readiness, ensuring that every process, from design to delivery, is audit-ready at all times.
What are the primary quality certifications required for medical equipment in US government tenders?
The primary certifications required are ISO 13485, FDA approval or clearance, and TAA compliance. ISO 13485 establishes the quality management system foundation, FDA ensures safety and efficacy for marketed devices, and TAA verifies that products originate from designated countries under U.S. federal procurement rules. These certifications are non-negotiable for eligibility and must be actively maintained with documented evidence.
What is the significance of ISO 13485 for medical device manufacturers participating in government tenders?
ISO 13485 is the globally accepted standard for quality management systems in medical device manufacturing and is increasingly adopted by regulators like the FDA as the basis for their quality system legislation. It demonstrates that a manufacturer has implemented a robust, risk-based system for design, production, and post-market surveillance. Government procurement teams prioritise suppliers with ISO 13485 certification because it reduces verification burden and signals operational maturity.
How can organisations ensure TAA compliance for medical equipment supplied to federal agencies?
Organisations must verify that all components of their medical equipment originate from countries designated under the Trade Agreements Act, typically including U.S. allies with reciprocal trade agreements. This requires mapping the entire supply chain, obtaining certificates of origin from suppliers, and maintaining audit-ready records. Automated compliance platforms can streamline this by integrating supplier data with TAA country lists and flagging non-compliant components before bid submission.